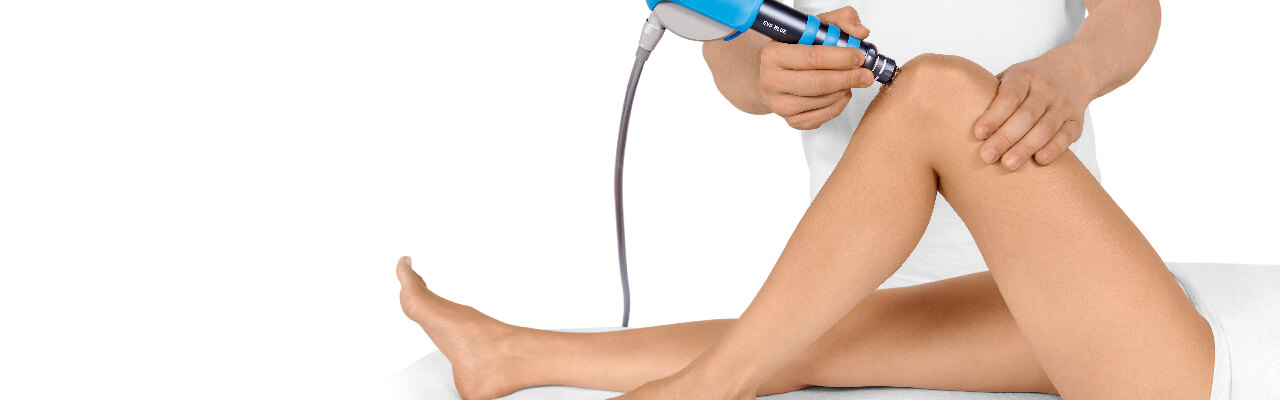
PATELLA TIP SYNDROME
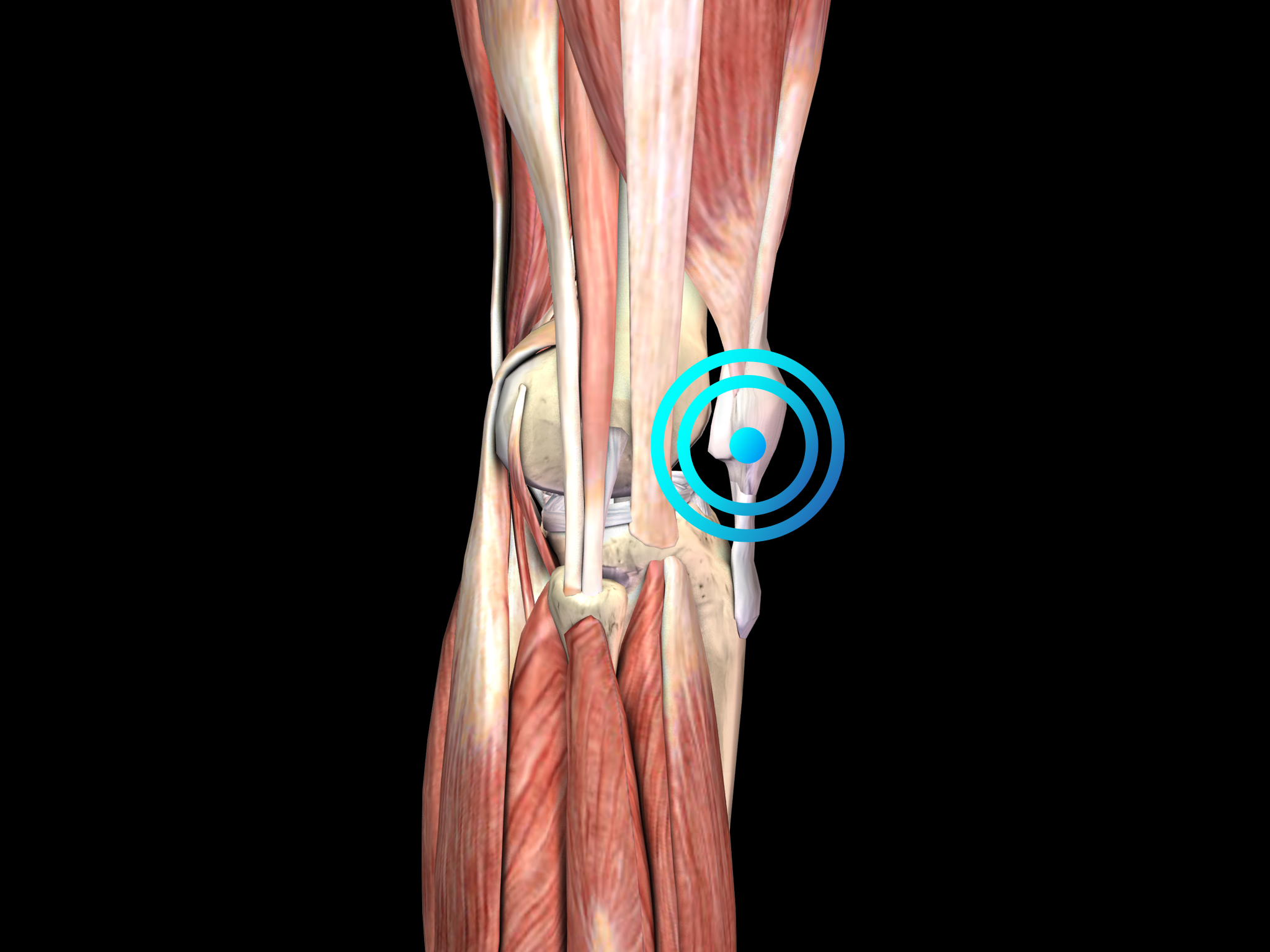
Pathology
The main clinical symptom is pain at the inferior pole of the patella. Diagnosis is based on the clinical features of the disease. Diagnostic imaging should be considered to rule out other causes of knee pain or to establish the diagnosis of patella tip syndrome when in doubt.
Similar to other tendinopathies, the etiology of patella tip syndrome is not completely understood, but repetitive overload is thought to be an important factor. Histologic examination of biopsy specimens from patients undergoing patellar tendon surgery for chronic symptoms has shown that chronic PT is associated with degenerative changes in the tendon. Accordingly, the disease is better characterized as “tendinopathy” than “tendinitis”, resembling the situation in other overuse tendon problems such as Achilles tendinopathy.
Athletes have a very high prevalence of jumper`s knee, i.e., up to 40% among elite basketball and volleyball players.
The condition can be debilitating and may prevent athletes from returning to sport for long periods between 6 months and more than 2 years.
The treatment of patella tip syndrome should start with conservative treatment modalities including rest, physiotherapy, eccentric strengthening, bracing and non-steroidal anti-inflammatory drugs.
Patients not responding to conservative treatment for six months should then undergo radial shock wave therapy for the treatment of patella tip syndrome.
Surgery should be considered for recalcitrant cases of patella tip syndrome. Numerous arthroscopic and open procedures have been described, but a consensus agreement about the best option is not available.
Side effects of Radial Shock Wave Therapy (RSWT) using the Swiss DolorClast®.
When performed properly, RSWT with the Swiss DolorClast® has only minimal risks. Typical device-related non-serious adverse events are:
- Pain and discomfort during and after treatment (anaesthesia is not necessary)
- Reddening of the skin
- Petechia
- Swelling and numbness of the skin over the treatment area
These device-related non-serious adverse events usually disappear within 36h after the treatment.
Treatment Procedure
Clinical Proof
Eccentric loading plus radial shock wave therapy in the treatment of chronic patellar tendinopathy.
Crupnik J.
Abstracts 12th International Congress of the International Society for Musculoskeletal Shockwave Therapy, Sorrento, Italy, 2009, 26-27
Treatment of patellar tendinopathy in combination with extracorporeal shock wave therapy with eccentric loading.
View Study →
Achillodynie und Patellaspitzensyndrom – Ergebnisse der Behandlung austherapierter, chronischer Fälle mit radialen Stosswellen.
Lohrer H, SchÖll J, Arentz S
Sportverl Sportschad 2002; 16: 108-114
"Achillodynia and patellar tip syndrome - results of the treatment of fully treated chronic cases with radial shock waves." (Study in German)
Recommended Settings
| Recommended Settings | Treatment | Myofascial Therapy |
| Number of treatment sessions | 3 to 5 | 3 to 5 |
| Interval between two sessions | 1 week | 1 week |
| Air pressure Evo Blue® | 1.5 to 3 bar | 3 to 4 bar |
| Air pressure Power+ | Not recommended | Not recommended |
| Impulses | 2000 on the painful spot |
2000
|
| Frequency | 8Hz to 12Hz | 12Hz to 20Hz |
| Applicator | 15mm | 36mm |
| Skin pressure | Light | Light to moderate |
Contraindications
The following contraindications of RSWT using the Swiss DolorClast® must be considered:
- Treatment over air-filled tissue (lung, gut)
- Treatment of pre-ruptured tendons
- Treatment of pregnant women
- Treatment of patients under the age of 18 years (except for Osgood-Schlatter disease and muscular dysfunction in children with spastic movement disorders)
- Treatment of patients with blood-clotting disorders (including local thrombosis)
- Treatment of patients treated with oral anticoagulants
- Treatment of tissue with local tumors or local bacterial and/or viral infections
- Treatment of patients treated with cortisone
Some indications may not be approved in the United States of America, under regulation by the US FDA. Please refer to the respective Instructions for Use.
